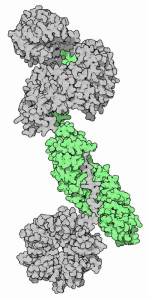
An Influenza virus is typically about the size of 70 millimicrons or a thousandth the width of a single strand of spider-web. To reproduce, viruses should be able to pass on their genetic message and often an enzyme to translate that message, the viral polymerase. The polymerase enzyme is crucial in bringing single monomers or nucleotides of DNA together, catalysing the polymerisation of DNA when a gene is copied. The conformation of the enzyme, or it’s shape, is crucial to it’s function. The virus, needing the host cell’s metabolism to survive, targets the cell’s nucleus. Isolated from the rest of the cell by the nuclear membrane, the nucleus is an ideal site for the replication mechanism. The gatekeeper proteins such as “importin ∝ ” are selective about the molecules that make it within the nucleus. In the case of influenza h5n1 researchers at The Institut de Biologie Structurale have been able to show a previously unknown conformation to the viral polymerase. This alternate conformation allows the polymerase to reach within the nucleus, attaching itself to the importin ∝ . It then has to acquire the conformation to be functional as the polymerase, replicating the viral message. What is interesting is that the polymerase’s conformational change is that it is dependent on a change in the temperature of it’s medium. A change in temperature can now be linked with a change in the polymerase’s efficiency. This could be why there are different rates of mutations of Influenza H5N1 within it’s avian and mammalian hosts.
F. Tarendeau, J. Boudet, S. Cusack, D.J. Hart et al. Structure and nuclear import function of the C-terminal domain of influenza virus polymerase PB2 subunit, Nature Structural and Molecular Biology, 25 February 2007